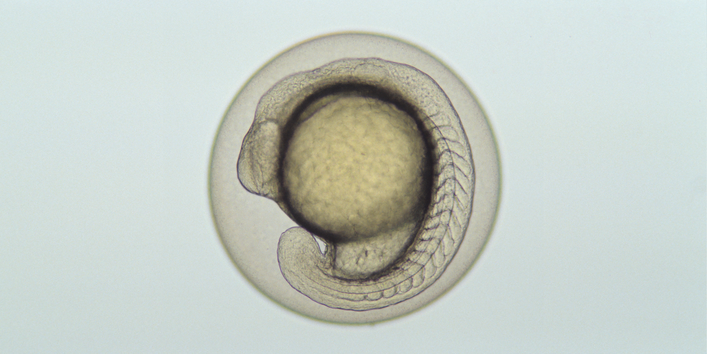
This report summarises the outcome of the expert workshop on the potential regulatory application of OECD Test Guideline 236 Fish Embryo Acute Toxicity Test (FET, OECD TG 236) under the REACH Regulation (No 1907/2006) , the Regulation on CLP (No 1272/2008) and the BPR (528/2012) held by European Chemical Agency (ECHA) and the German Environment Agency (Umweltbundesamt, UBA) at the premises of ECHA in Helsinki on 3rd and 4th May 2017.
The workshop participants were composed of experts from academia, industry and governmental organizations as well as non-governmental organizations (NGOs). The list of participants is provided in Annex 1.
The workshop was jointly organised by ECHA and UBA, with support of UBA Austria and EURL ECVAM in the steering committee.
The aim of the workshop was to exchange views on the potential regulatory application of the FET and explore possibilities on how the FET might be used as a part of weight of evidence approaches in the EU regulatory context (REACH, BPR and CLP) to adapt standard information requirements for acute fish toxicity. Under REACH, information on short-term toxicity fish is standard requirement for all substances manufactured or imported in the EU in quantities of 10 tonnes or more. This information is used in risk assessment and identification of Persistent, Bioaccumulative and Toxic substances (PBT assessment) under the REACH Regulation and for hazard classification under the Regulation on CLP.
Background to the workshop is the OECD TG 236 Fish Embryo Acute Toxicity (FET, OECD TG 236) developed under the lead of Germany and approved in 2013. OECD TG 236 was developed and validated (OECD Series on Testing and Assessment No. 157 and 179 Validation Reports Phase 1 and 2 for the Zebrafish Embryo Toxicity Test) with the goal to determine acute toxicity of chemicals on embryonic fish on the basis of a positive outcome in any of the four indicators of lethality recorded, and to calculate the LC50. The acute embryo toxicity has been shown to correlate well with the acute adult fish toxicity and therefore the FET test (OECD TG 236) may be a promising alternative to standard Fish Acute Toxicity (AFT) Test (OECD TG 203).
As the FET test is not set out as an alternative method to adapt standard information requirement for short-term toxicity to fish in the REACH Regulation, in 2015 ECHA contracted out the analysis of relevance and adequateness of the new test (OECD TG 236) under the REACH Regulation. The results of this project were discussed with experts from the Member State competent authorities during 2015. The report of this work was published on ECHA’s website as ‘Analysis of the relevance and adequateness of using Fish Embryo Acute Toxicity test (FET) Test Guideline (OECD TG 236) to fulfil the information requirements and addressing concerns under REACH’ (link) together with official recommendation of ECHA on how to use the method under REACH. From the report ECHA concluded that the FET could not be considered as a stand-alone information for adapting the information requirement for the acute fish toxicity test under the REACH Regulation. Based on current knowledge, ECHA considered that OECD TG 236 may be used within a weight of evidence approach (Annex XI, Section 1.2 to the REACH Regulation) together with other independent, adequate, relevant and reliable sources of information leading to the conclusion that the substance has or does not have a particular dangerous property.
Moreover, it has to be noted that at OECD level, Austria and ICAPO (International Council for Animal Protection in OECD Programmes) are leading the OECD project no. 2.54 on inclusion of the FET into OECD Guidance Document No. 126 “Short Guidance on the Threshold Approach for Acute Fish Toxicity” (OECD GD 126). Discussion on the revision is ongoing.
Fish Embryo Acute Toxicity (FET) test: workshop report available
UBA and the European Chemical Agency (ECHA) organised an expert workshop in May 2017. The aim was to exchange views on the potential applicability of the FET test for regulatory purposes under REACH, Biocidal Products- and Classifcation and Packaging Regulation and explore how it might be used as part of weight of evidence approaches for acute fish toxicity.